7 Dosage Forms Available
Every Custom Formula, Any Dosage Form
Your custom nutraceutical formulation can be manufactured in any of the 7 dosage forms we produce in our WHO-GMP facility. Choose the format that best suits your market positioning and consumer preference.
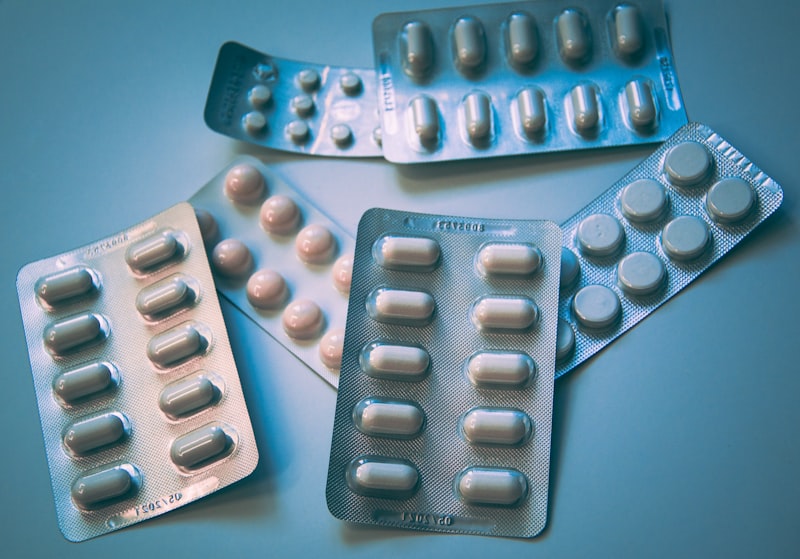
Capsules
Vegetarian HPMC and gelatin capsules. Sizes 00 to 4. Custom fill weights and blends.
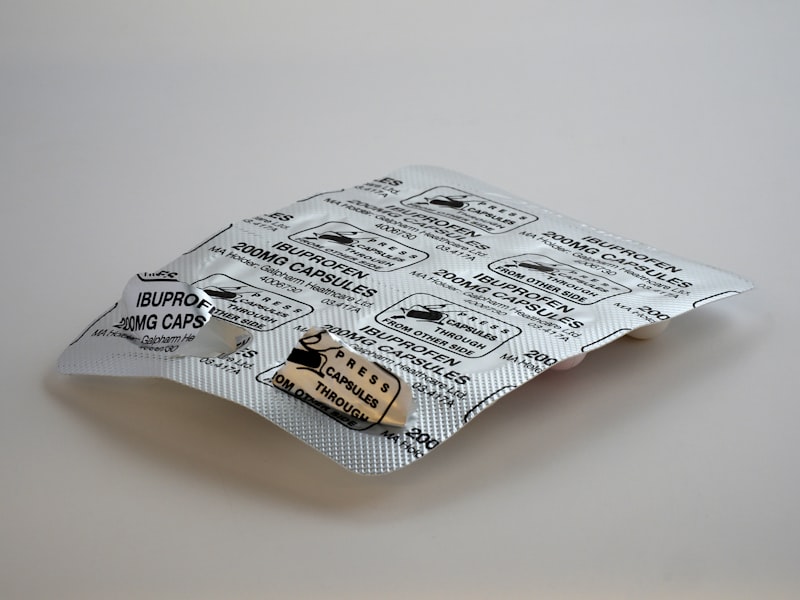
Tablets
Film-coated, chewable, effervescent, and sustained-release tablets. Custom shapes and sizes.

Softgels
Oil-based and liquid-fill softgel capsules. Ideal for fat-soluble vitamins, omega-3s, and CoQ10.
Syrups
Liquid suspensions and syrups. Custom flavoring, sugar-free options, and pediatric formulations.

Powders
Protein blends, superfood mixes, and functional powders. Custom particle size and flavoring.

Sachets
Single-dose stick packs and sachets. Granules, powders, and effervescent formulations.

Ayurvedic
AYUSH GMP certified. Classical and proprietary Ayurvedic formulations with full traceability.